As part of The Periodic Table article series, this article serves as a revision of the previous article, and provides more extensive detail compared to the previous.
Overview
| Property | Value |
| Symbol | H |
| Atomic Number | 1 |
| Atomic Mass | 1.008 u |
| Group | 1 |
| Period | 1 |
| Classification | Non-Metal |
| Electron Configuration | 1s¹ |
| State at 25°C | Gas |
Hydrogen is the simplest and most abundant element in the universe, accounting for approximately 75% of its elemental mass. [1]
It was formed within the first few minutes following the Big Bang, during a process known as the Big Bang nucleosynthesis, when temperatures and densities allowed protons to combine with electrons for the first time. [2]
This elemental simplicity of one proton and one electron do not fully capture the extent of hydrogen’s extraordinary importance.
Hydrogen fuels stars through nuclear fusion, forms water through chemical bonding, and underpins all known biological molecules.
In modern times, it is also a central point to discussions of clean and sustainable energy systems. [3]
Discovery and Origin
Although hydrogen gas was produced unknowingly for centuries, it was first identified as a distinct substance in 1766 by English scientist Henry Cavendish, who referred to it as “inflammable air”. [4]
Cavendish demonstrated that burning this gas produced water, a finding later interpreted by Antoine Lavoisier, who named the element hydrogène (Greek for “water former”). [5]

Henry Cavendish (left) and Antoine-Laurent Lavoisier (right) — 18th-century pioneers of modern chemistry. Cavendish identified hydrogen as a distinct substance, while Lavoisier named the element and established the principles that shapes modern chemical science.
This discovery played a foundational role in dismantling the phlogiston theory and establishing modern chemical nomenclature.
Atomic Structure and Isotopes
Hydrogen has the simplest atomic structure of any element:
- 1 proton
- 1 electron
- 0 neutrons (in its most common form)
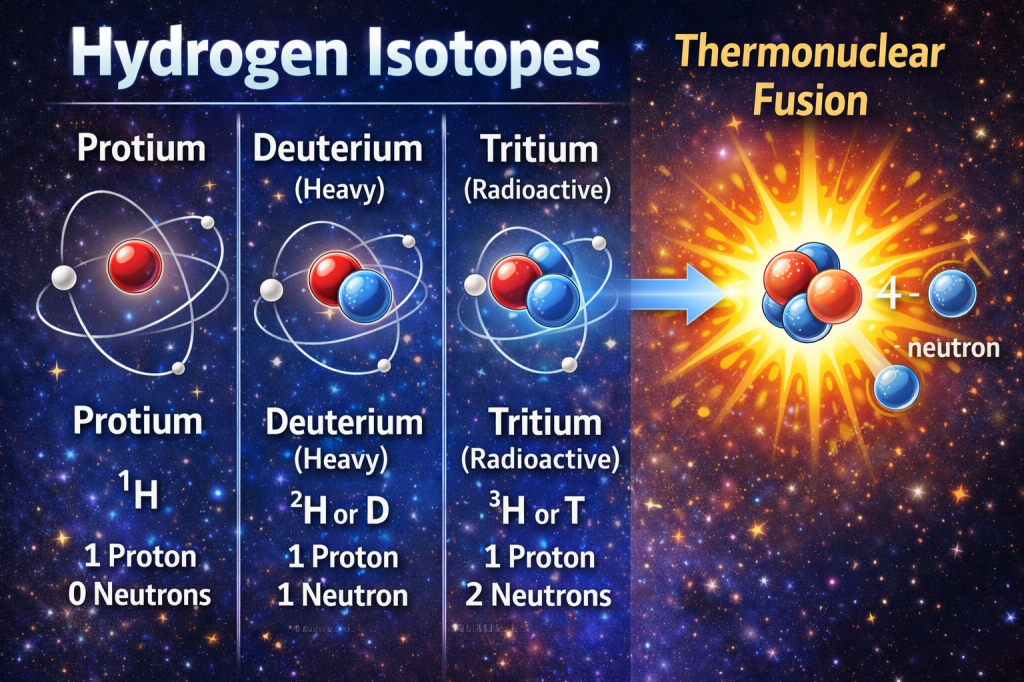
Despite this simplicity, hydrogen exists in three naturally occurring isotopes:
| Isotope | Symbol | Neutrons | Notes |
|---|---|---|---|
| Protium | ¹H | 0 | ~99.98% abundance |
| Deuterium | ²H (D) | 1 | Used in heavy water and nuclear research |
| Tritium | ³H (T) | 2 | Radioactive; used in fusion research |
Deuterium and tritium are especially important in thermonuclear fusion, the same process that powers stars and experimental fusion reactors on Earth. [6]
Natural Occurrence and Production
Natural Occurrence
Hydrogen is rarely found freely as a gas on Earth due to its low atomic mass and high reactivity. Instead, it is bound in compounds such as:
- Water (H₂O)
- Hydrocarbons (E.g. natural gas, petroleum)
- Organic molecules in all living organisms
In contrast, hydrogen dominates the composition of stars, nebulae, and gas giants like Jupiter. [7]
Industrial Production
Currently, most hydrogen is produced via:
- Steam Methane Reforming (SMR) – fossil fuel based (non-renewable)
- Electrolysis of water – water based (renewable)
- Biological and photochemical methods – emerging technologies
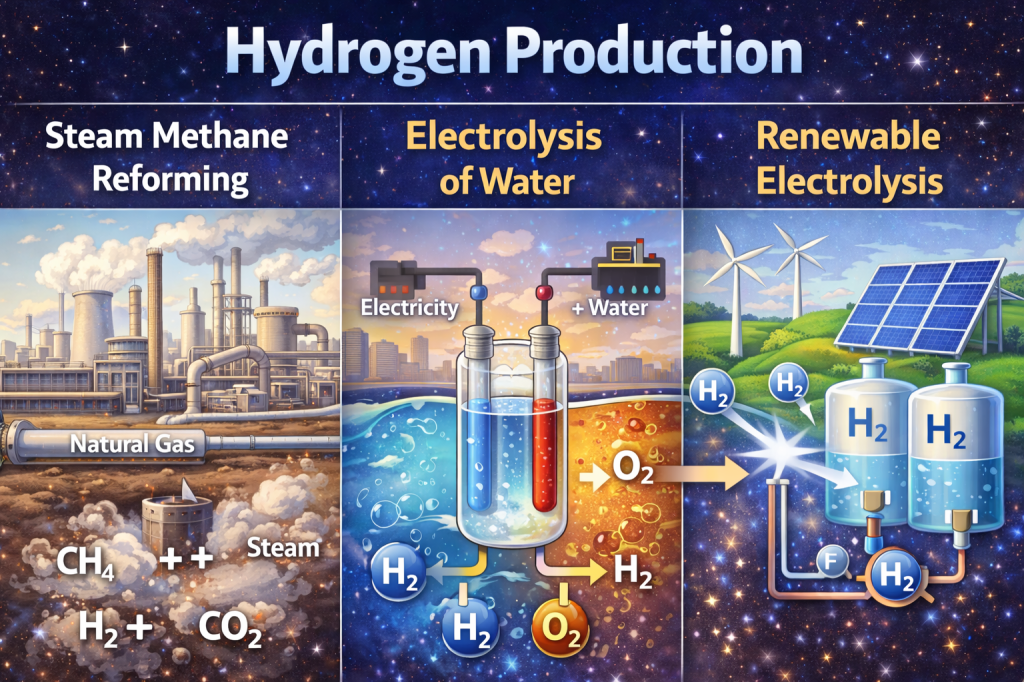
Green hydrogen, produced using renewable electricity, is a major focus of climate mitigation strategies. [8]
Application and Uses
Energy and Fuel Cells
Hydrogen fuel cells generate electricity through electrochemical reactions, emitting only water vapor as a by-product.
These systems are used in:
- Hydrogen-powered vehicles
- Backup power systems
- Space missions
Fuel cells are considered among the most promising technologies for decarbonizing transportation and heavy industry. [9, 10]
Industrial Chemistry
Hydrogen is essential for:
- Ammonia synthesis (Haber–Bosch process)
- Petroleum refining
- Methanol and polymer production
Over 50% of global hydrogen production is used in fertilizer manufacturing alone. [11]
Aerospace and Research
Liquid hydrogen is widely used as rocket fuel due to its high energy density per unit mass. [12]
Biological and Environmental Role
Hydrogen is a fundamental component of all biological molecules, including:
- Carbohydrates
- Proteins
- Lipids
- DNA and RNA
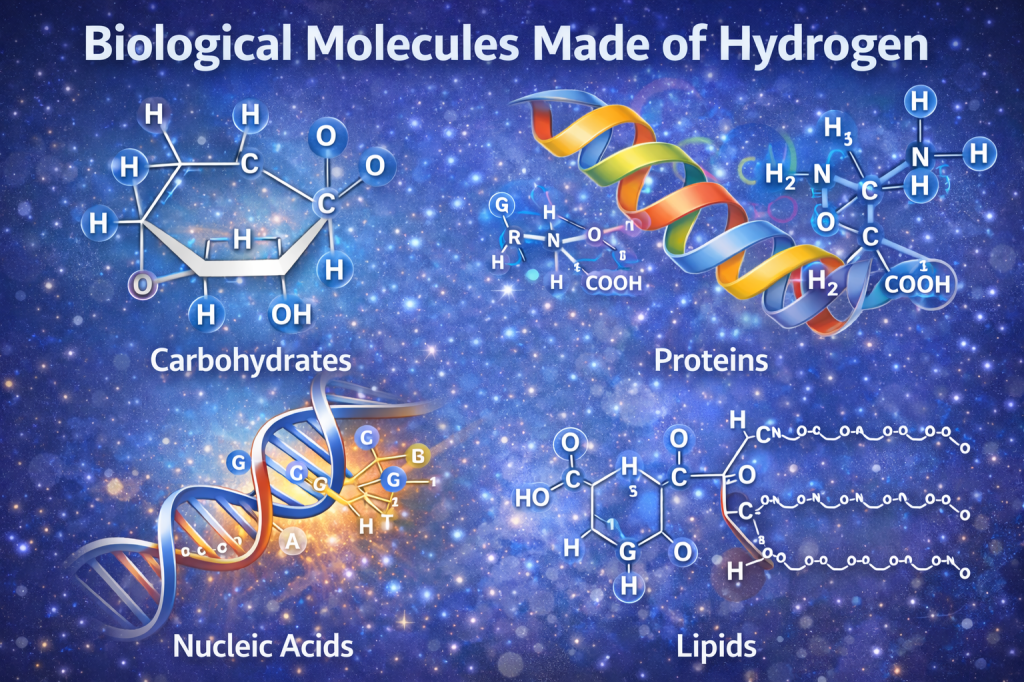
Hydrogen bonds play a critical role in:
- Protein folding
- Enzyme specificity
- DNA base pairing
In environmental science, hydrogen cycling influences atmospheric chemistry and climate interactions. [13]
Safety and Hazards
Hydrogen gas is:
- Highly flammable
- Colourless and odourless
- Explosive when mixed with air at certain concentrations
Historical incidents such as the Hindenburg disaster (1937) highlighted these risks. Modern hydrogen systems, however, incorporate advanced materials, sensors, and safety standards that significantly reduce danger. [14]
Scientific and Cultural Significance
- Hydrogen was first detected in the Sun’s spectrum before being identified on Earth [15]
- It is the only element that does not fit neatly into any single periodic table group.
- The term “hydrogen economy” refers to future energy systems built around hydrogen as a primary fuel.

Hydrogen remains central to research in cosmology, chemistry, biology, and energy science.
Related Elements
- Helium (He): Formed by hydrogen fusion in stars
- Oxygen (O): Combines with hydrogen to form water
- Carbon (C): Forms hydrocarbons with hydrogen
References
[1] Planck 2018 results – VI. Cosmological parameters (Corrigendum) | Astronomy & Astrophysics (A&A)
[2] Principles of Physical Cosmology | Princeton University Press
[3] Sustainable Hydrogen Production | Science
[5] Lavoisier’s “Reflections on phlogiston” I: against phlogiston theory | Foundations of Chemistry
[6] Handbook of Isotopes in the Cosmos
[7] An Introduction to the Theory of Stellar Structure and Evolution
[8] Global Hydrogen Review 2022
[9] Sustainable hydrogen production
[10] The role of hydrogen and fuel cells in the global energy system
[11] Enriching the Earth | Fritz Haber, Carl Bosch, and the Transformation of World Food Production
[12] Rocket Propulsion Elements, 9th Edition
[13] The tropospheric cycle of H2: a critical review
[14] Safety, Codes and Standards
[15] August 18 and October 20, 1868: Discovery of Helium
Authors Note: Eddy Xu is trying to express his interest in STEM and provide educated articles to allow readers from all ages to learn science and tech. He has a strong passion to develop new learning methods to bridge the gap between education and industry practice in his journey.
Leave a comment