As part of The Periodic Table article series, this article serves as a revision of the previous article, and provides more extensive detail compared to the previous.
Overview
| Property | Value |
| Symbol | He |
| Atomic Number | 2 |
| Atomic Mass | 4.0026 u |
| Group | 18 |
| Period | 1 |
| Classification | Noble gas |
| Electron Configuration | 1s² |
| State at 25°C | Gas |
Helium is the second most abundant element in the universe, accounting for approximately 24% of the universe’s elemental mass, formed primarily through Hydrogen fusion in stars. [1]
Unlike hydrogen, helium is chemically inert under most conditions, a property that places it among the noble gases.
Helium’s importance spans across cosmology, cryogenics, quantum physics, and medicine.

Though plentiful in the universe, helium is relatively scarce on Earth, making it both scientifically valuable and economically strategic. [2]
Discovery and Naming
Helium was first discovered in the Sun before it was found on Earth.
In 1868, astronomer Jules Janssen and Sir Norman Lockyer independently observed a previously unknown yellow spectral line during a solar eclipse. [3]

Lockyer proposed the name helium, derived from Helios, the Greek god of the Sun.
Helium was not isolated on Earth until 1895, when Sir William Ramsay detected it in uranium-bearing minerals, confirming its terrestrial presence. [4]
This marked the first discovery of an element beyond Earth before its identification on the planet.
Atomic Structure and Isotopes
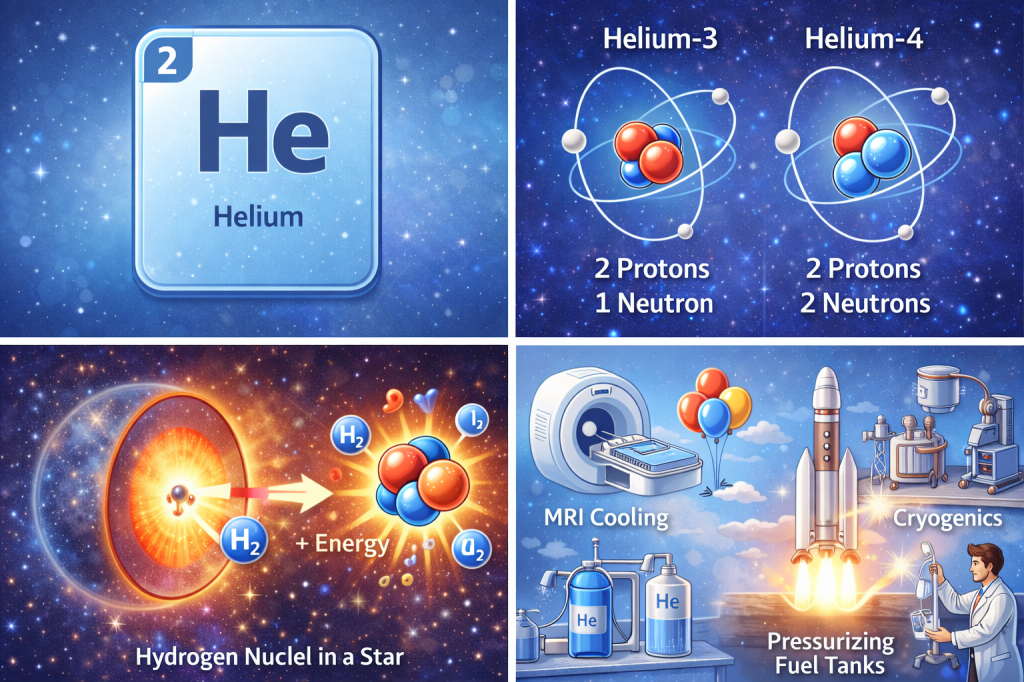
Helium atoms consist of:
- 2 proton
- 2 electron
- Typically 2 neutrons
Helium has two stable isotopes:
| Isotope | Symbol | Neutrons | Notes |
|---|---|---|---|
| Helium-3 | ³He | 1 | Rare (on Earth); used in fusion research and neutron detection |
| Helium-4 | ⁴He | 2 | ~99.98% abundance |

Helium-4 nuclei, also known as alpha particles, are produced during radioactive decay and stellar fusion processes. [5]
Helium-3 is known to be abundant on the surface of the moon, and is a vital strategic resource for the recent developments of nuclear fusion reactors.

Natural Occurrence and Formation
Cosmic Origin
Helium is formed through:
- Big Bang nucleosynthesis
- Stellar hydrogen fusion
- Radioactive decay of heavy elements
In stars, hydrogen nuclei fuse to form helium through the proton–proton chain reaction, releasing vast amounts of energy. [6]
Terrestrial Sources
On Earth, helium accumulates underground as a decay product of uranium and thorium. It is typically extracted as a by-product of natural gas processing. [2]
Application and Uses
❄️ Cryogenics and Superconductivity
Liquid helium has the lowest boiling point of any element (4.2 K), making it indispensable for:
- MRI scanners
- Particle accelerators
- Superconducting magnets
Helium-4 enables superconductivity by maintaining extremely low temperatures required for quantum phenomena [7].
🧠 Scientific Research
Helium is essential in:
- Quantum fluid studies (superfluidity)
- Low-temperature physics
- Neutron detection (³He detectors)
Superfluid helium exhibits zero viscosity, challenging classical physics models. [8]
🚀 Aerospace and Industry
Helium is used to:
- Pressurize rocket fuel tanks
- Purge fuel lines
- Leak-test high-precision systems
Its chemical inertness makes it uniquely safe for these applications. [9]
Biological and Environmental Role
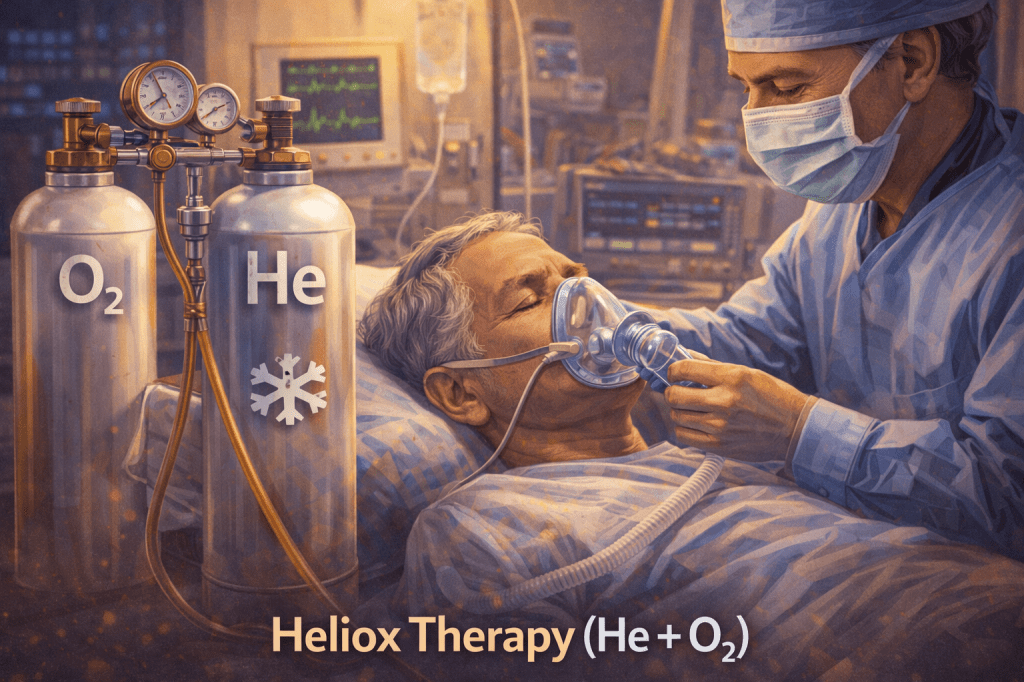
Helium is biologically inert and does not participate in biochemical reactions. However, it has medical applications in:
- Heliox therapy (helium–oxygen mixtures) to improve airflow in respiratory distress. [10]
Helium does not contribute to greenhouse effects or atmospheric chemistry due to its non-reactive nature.
Safety and Hazards
Although non-toxic and non-flammable, helium poses risks due to:
- Oxygen displacement (asphyxiation hazard)
- High-pressure storage
Global helium shortages have raised concerns over sustainability, recycling, and strategic reserves. [11]
Scientific and Cultural Significance
- Helium was the first element discovered extraterrestrially
- It validated stellar nucleosynthesis theory
- It is essential to modern medical imaging and quantum research
Despite its common association with balloons, helium is one of the most scientifically valuable elements on Earth.
Due to its scarcity, excessive uses for cosmetic purposes should be taken caution, as continued commercial sale for consumers to use for entertainment can cause resources to dwindle further resulting in the destruction of available reserves for future research.

Related Elements
- Hydrogen (H): Fuses to form helium in stars
- Neon (Ne): Next noble gas in the periodic table
- Lithium (Li): Formed after helium in stellar evolution
References
[2] THE COMPOSITION OF- THE EARTH’S CRUST
[3] Spectroscopic Observations of the Sun
[4] On a Gas Showing the Spectrum of Helium
[5] Handbook of Isotopes in the Cosmos
[6] Solar fusion cross sections. II. The 𝑝𝑝 chain and CNO cycles
[7] Handbook on the Physics and Chemistry of Rare Earths
[8] Quantum Liquids
[9] Rocket Propulsion Elements, 9th Edition
[10] The Applications of Heliox therapy, article review
[11] The Future of Helium as a Natural Resource
Authors Note: Eddy Xu is trying to express his interest in STEM and provide educated articles to allow readers from all ages to learn science and tech. He has a strong passion to develop new learning methods to bridge the gap between education and industry practice in his journey.
Leave a comment