Background
I was engaged as the technical lead to resolve a critical and escalating quality failure affecting a long-established wax-based semi-solid medicinal product containing an active pharmaceutical ingredient (API).
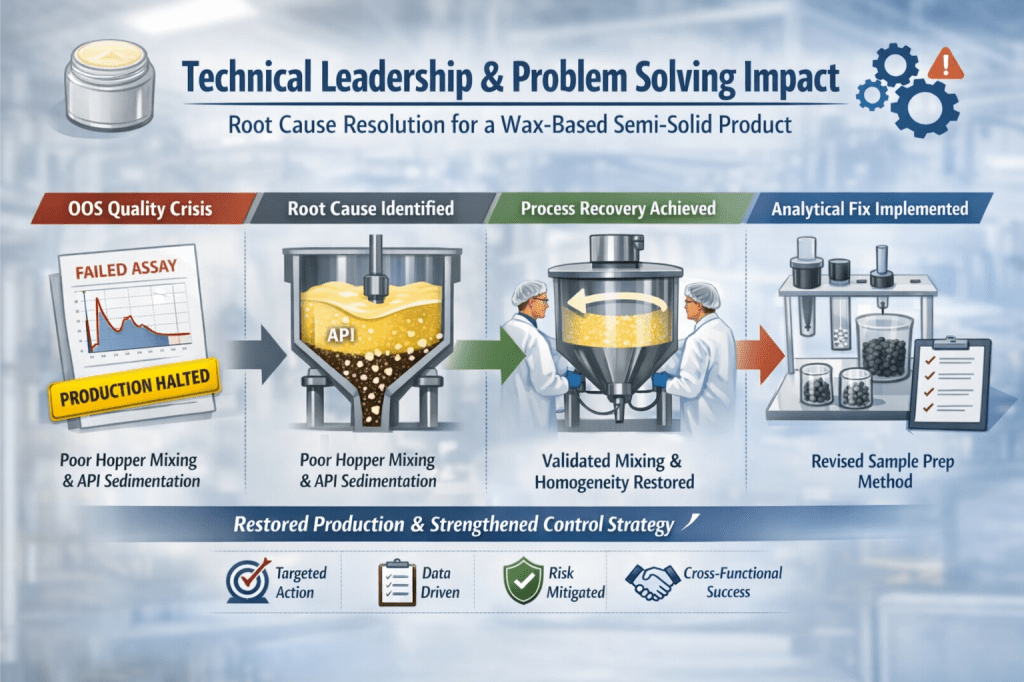
After years of stable commercial manufacture, multiple consecutive batches began failing API assay specifications.
Failures were systemic, appearing in composite samples as well as at early and mid-fill stages, indicating a loss of process control rather than isolated sampling error.
The issue triggered a production shutdown, creating a high-risk supply interruption for a critical medicine and increasing the likelihood of regulatory escalation.
The situation required rapid technical direction, disciplined investigation, and confident decision-making under operational and compliance pressure.
Leadership-Led Investigation Strategy
I led the root cause investigation using the site’s formal RCA framework aligned with Lean principles, bringing together Manufacturing, Technical Services, and Quality into a single, focused investigation team.
My role was to structure the investigation, challenge assumptions, and ensure evidence-based decision-making rather than reactive troubleshooting.
Initial reviews across five failed batches systematically eliminated raw material variability, operator practices, and procedural non-compliance as causal factors.
The repeatable nature of the failures pointed to a latent design or process weakness rather than execution error.
I redirected the investigation toward the intermediate bulk handling and mixing configuration during the filling phase — an area previously considered low risk due to historical performance.
Experimental Direction and Root Cause Confirmation
Recognising that theoretical analysis alone would not resolve the issue, I led the technical risk assessment and successfully obtained approval to execute a tightly controlled trial batch under cross-functional oversight.
This decision balanced patient supply risk against the need for decisive data.
During the trial, I personally oversaw bulk transfer behaviour, hopper agitation performance, and temperature control throughout the filling sequence.
Real-time observations revealed that the intermediate hopper stirrer was insufficient to maintain homogeneity in the wax–API matrix, allowing API sedimentation during hold and discharge.
This hypothesis was confirmed when significant assay variation was detected between palletised units sampled during unloading of a 20 kg bulk.
To conclusively demonstrate causality, I designed and executed a controlled intervention: the same bulk was reintroduced into the hopper and mixed at a defined speed of 200 rpm prior to filling.
All subsequent samples met specification with uniform API distribution, definitively establishing inadequate hopper mixing as the primary root cause.
Extending the Investigation Beyond the Obvious
While process control was restored, I identified that assay variability between duplicate laboratory results remained unacceptably high.
Rather than accepting this as analytical noise, I expanded the scope of the investigation to include laboratory practices — a critical leadership decision that prevented recurrence through a different failure mode.
My review of the analytical method identified a hidden weakness in the sample preparation step.
The existing procedure required melting and hand-mixing 20 pellets to form a composite sample.
This practice unintentionally promoted API demixing within the molten wax, directly undermining assay reproducibility.
I led the redesign of the sample preparation approach to eliminate melting entirely, instead analysing intact pellets.
The revised method was formally revalidated and demonstrated significantly improved precision, accuracy, and repeatability.
Outcome and Impact
Through decisive technical leadership and structured problem-solving, I restored control of a complex semi-solid manufacturing process and prevented prolonged product unavailability.
Key outcomes included:
- Identification and correction of a latent design weakness in intermediate hopper mixing.
- Implementation of a robust in-process agitation strategy ensuring consistent API distribution.
- Redesign and revalidation of a flawed analytical sample preparation method.
- Restoration of commercial supply with strengthened process and analytical control.
- Enhanced cross-functional confidence in the site’s ability to manage complex formulations.
This intervention not only resolved the immediate crisis but materially strengthened the site’s technical capability, investigation maturity, and control strategy for semi-solid dosage forms — demonstrating the value of strong technical leadership in high-risk manufacturing environments.
Authors Note: Wilson Prasad also known as user name muefatiaki1966 is trying to leverage his extensive experience within the industry to educate and invoke discussion for topics of interest.
Leave a comment