The Accidental Discovery of Exosomes
Exosomes were not discovered because scientists were searching for a new class of biological particles.
Instead, they emerged from a much more practical question:
How do cells get rid of unwanted components as they mature?
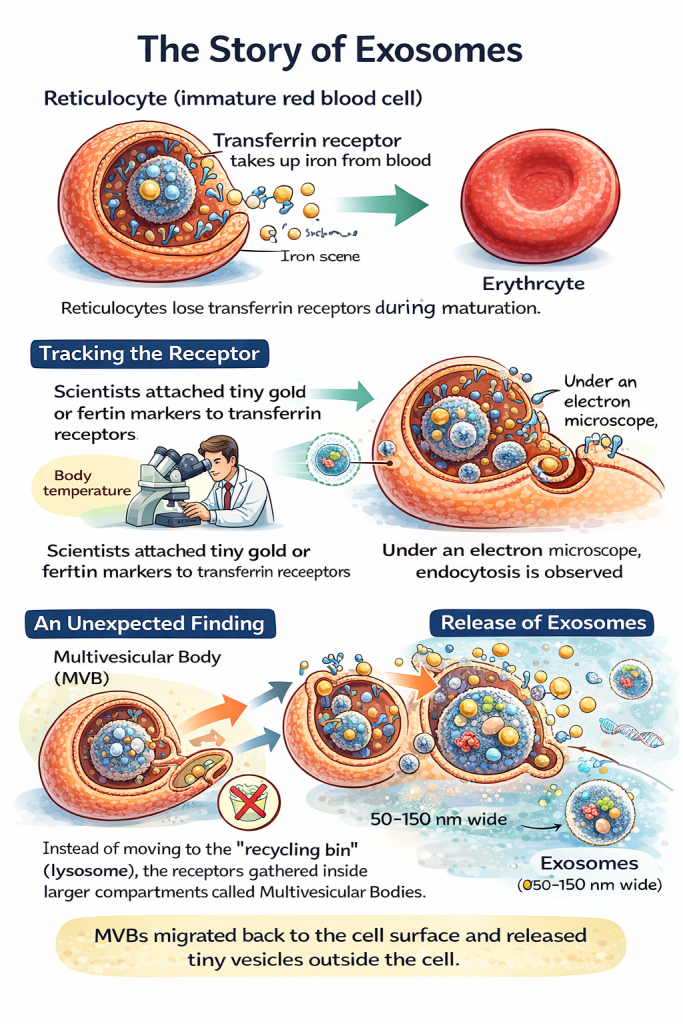
The story begins with reticulocytes, immature red blood cells.
As reticulocytes mature into erythrocytes, they lose many surface proteins they no longer need.
One of these proteins is the transferrin receptor, which is responsible for iron uptake from the bloodstream.
Once a red blood cell is fully mature, iron uptake is no longer required—so the transferrin receptor must be removed.
In the early 1980s, exactly how this happened was still unknown.
Following the Transferrin Receptor
In 1983, research groups led independently by Stahl and Johnstone set out to solve this mystery.
To track the fate of the transferrin receptor, researchers tagged it with tiny, electron-dense markers made from gold particles or ferritin.
These markers allowed the receptors to be followed under powerful electron microscopes.
What they observed was striking:
- At low temperatures, the transferrin receptors remained on the cell surface.
- At physiological (body) temperature, the receptors were pulled into the cell inside small pockets of membrane.
These pockets pinched off from the surface, forming membrane-bound vesicles that travelled inward—classic receptor-mediated endocytosis.
So far, everything matched expectations.
An Unexpected Cellular Detour
The real surprise came next.
Instead of being delivered to the cell’s degradation machinery ( called the lysosome, often described as the cell’s recycling bin), the receptors accumulated inside much larger compartments filled with many tiny vesicles.
These structures are now known as multivesicular bodies (MVBs).
Each MVB contained dozens to hundreds of small internal vesicles, formed by inward budding of the endosomal membrane.
At the time, no one knew what these structures were for.
Release Instead of Recycling
Here came the final and most unexpected observation.
Rather than fusing with lysosomes and degrading their contents, some multivesicular bodies migrated back to the cell surface.
When they fused with the plasma membrane, the tiny vesicles inside were released into the extracellular space.
Each of these vesicles was approximately 50 nanometres in diameter, far too small to be seen without electron microscopy.
At the time, researchers assumed this was simply an efficient disposal mechanism: a way for reticulocytes to shed unwanted receptors during maturation.
No one yet suspected that these vesicles might have a much bigger role.
From Cellular Waste to Biological Messengers
Years later, it became clear that these released vesicles were not inert debris.
They could:
- Travel to other cells.
- Be taken up by target cells.
- Deliver proteins, lipids, and RNA.
- Act as a form of intercellular communication.
These vesicles were eventually given a name:
“Exosomes“
What began as a study of iron uptake and receptor recycling turned out to reveal an entirely new biological communication system.
Exosomes Today
Today, exosomes are no longer viewed as cellular waste.
They are recognised as powerful biological messengers involved in:
- Immune regulation
- Cancer progression and diagnostics
- Tissue regeneration
- Neurobiology
- Targeted drug delivery
Because exosome membranes closely resemble those of their parent cells, they are readily recognised and internalised by recipient cells.
This property has made them one of the most actively studied platforms for next-generation therapeutics and precision medicine.
All of this unexpectedly began with a simple question about how a maturing red blood cell throws things away.
References
Harding, C. V., Heuser, J. E., & Stahl, P. D. (1983). Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes.. Journal of Cell Biology, 97(2), 329-339. https://doi.org/10.1083/jcb.97.2.329
Pan, B. T., Teng, K., Wu, C., Adam, M., & Johnstone, R. M. (1985). Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes. Journal of Cell Biology, 101(3), 942–948. https://doi.org/10.1083/jcb.101.3.942
Harding, C. V., Heuser, J. E., & Stahl, P. D. (2013). Exosomes: Looking back three decades and into the future. Journal of Cell Biology, 200(4), 367–371. https://doi.org/10.1083/jcb.201212113
Edgar, J. R. (2016). Q&A: What are exosomes, exactly? BMC Biology, 14, 46. https://doi.org/10.1186/s12915-016-0268-z
Authors Note: Niki Kakkad also known as user name nitikakkad2023 is trying to educate and invoke discussion on topics that she finds fascinate about.
Leave a comment