Continuing on from the previous article.
If Pseudoephedrine (PSE) is to return in a broader, clinically meaningful way in Australia, it won’t happen through opinion, nostalgia, or market pressure.
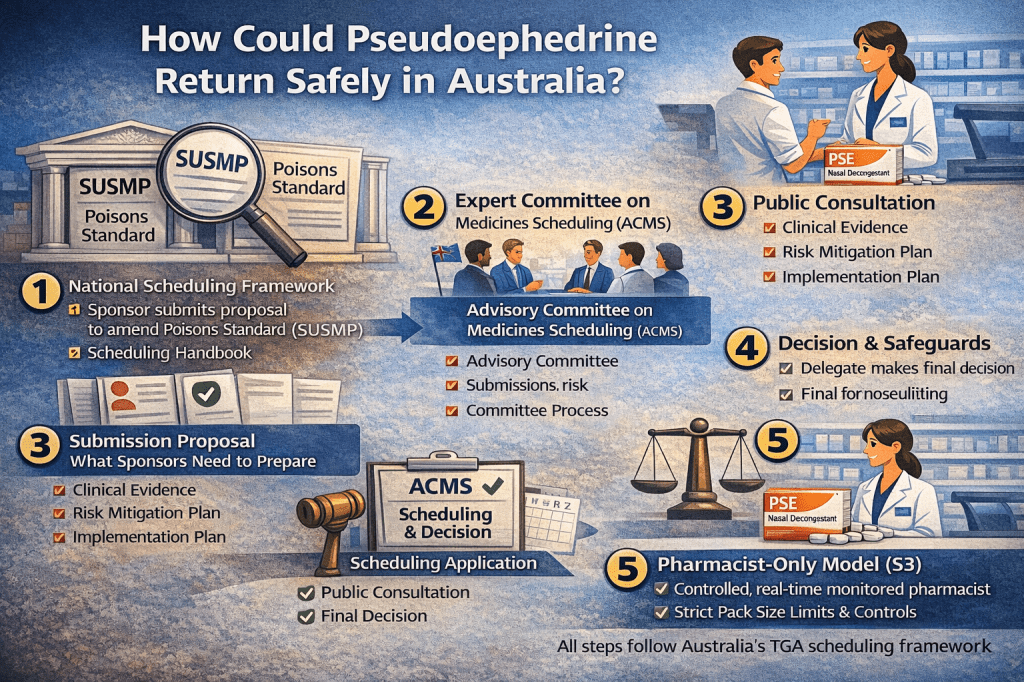
It will happen through the scheduling frameworks:
- The Poisons Standard (SUSMP) [1],
- The Scheduling Policy Framework [2],
- The Scheduling Handbook [3],
- and the expert advice and critical decision processes surrounding the Advisory Committee on Medicines Scheduling (ACMS) [4].
This article maps the real step by step pathway, so the conversation can move from “should we?” to “how could we, responsibly?”
The Foundation: What “Scheduling” means in Australia?
Australia’s Poisons Standard (also called the SUSMP) is the national legislative instrument that promotes uniform scheduling, labelling, and packaging controls for medicines and poisons across jurisdictions. [1]
While enforcement is implemented through state and territory laws, the SUSMP is the national reference point that creates practical consistency. [5, 6]
Scheduling is not just categorisation. It is risk management, for the determination of:
- Who can access a medicine
- Under what conditions
- With what safeguards (e.g., pharmacist oversight, warning statements, pack limits)
Australia’s scheduling system and the decision process are guided by the Scheduling Policy Framework and explained in operational detail in the Scheduling Handbook. [2, 3]
Who decides: the delegate and the ACMS?
Scheduling decisions are made by the Secretary (or delegate) under the Therapeutic Goods Act framework.
Key public health matters the decision-maker must consider are set out in legislation (for example, risks and benefits, purpose of use, toxicity, dosage, potential for abuse, and other public-health impacts). [6]
The ACMS is an expert advisory body that provides advice to the decision-maker (the delegate) on medicines scheduling proposals and considerations.
The TGA publishes ACMS information, including its role and meeting schedules/timeframes. [4, 7]
A practical way to describe it:
- the ACMS advises (clinical, public health, risk controls)
- the delegate decides (interim and final decisions under the scheduling process)
What actually triggers change: an Application or a Delegate-initiated proposal?
There are two common pathways to scheduling changes:
- Application to amend the Poisons Standard (by a sponsor, organisation, professional body, or other stakeholder) [3, 7]
- Delegate-initiated proposal (where the regulator initiates a review based on emerging evidence, signals, or policy concerns)
The TGA’s scheduling process runs through structured stages, including:
- Pre-meeting publication of proposed amendments
- Public consultation and submissions
- Committee consideration (ACMS / joint committee where relevant)
- Delegate interim decision (often with a further public submission stage)
- Delegate final decision and implementation timing
You can see how the consultation cycles and committee meeting processes operate via the TGA’s scheduling consultation pages and “Scheduling submissions” guidance. [8]
What a credible PSE proposal must contain?
For pseudoephedrine, an evidence-based proposal needs to do two things at once:
A) Demonstrate clinical need and benefit
This includes:
- Evidence that PSE provides measurable relief of congestion
- Evidence that current alternatives (notably oral phenylephrine) may not provide equivalent benefit
- Public-health implications of ineffective OTC pathways (e.g., downstream GP visits, inappropriate antibiotic requests, reduced confidence in OTC care)
Refer to this previous article for detailed scientific discovery of PSE and PE differences.
B) Provide a modern risk-control package
Any proposal that focuses only on efficacy will stall. The scheduling framework expects a sponsor to address risk management explicitly.
The Scheduling Policy Framework is designed to help decision-makers assess “best fit” scheduling with contemporary safeguards. [2, 3]
For PSE, the strongest risk controls are already established in Australia:
- Real-time recording at point of supply in jurisdictions such as NSW. [9]
- Pharmacist decision support tools such as Project STOP, which records PSE requests and supports refusal where diversion risk is identified. [10, 11]
- Professional guidance acknowledging Project STOP’s role in assisting supply decisions.
There is also current peer-reviewed research examining impacts of PSE regulation and monitoring approaches in Australian pharmacy contexts which are useful when framing “what worked, what didn’t, what improved over time”. [12]
Building the submission: the structure regulators expect
A high-quality scheduling submission typically includes:
1. The proposal (what change you want)
Examples might include (conceptually):
- Clarifying schedule entry language
- Setting pack-size / quantity limits
- Defining conditions of supply consistent with pharmacist oversight
- Aligning monitoring requirements with contemporary practice
The Pharmacy Guild has publicly discussed the importance of clear, unambiguous SUSMP entries, ideally using pack size / strength limitations where possible for useful framing when drafting controls. [13]
2. Evidence dossier (benefit and risk)
- Clinical efficacy evidence (PSE vs alternatives)
- Adverse effect profile and contraindications
- Misuse / diversion risk profile
- Real-world systems already mitigating diversion (Project STOP + jurisdiction requirements)
3. Risk mitigation plan (specific, auditable controls)
- Pharmacist screening and counselling
- Real-time recording and refusal protocols
- Storage/security expectations
- Audit KPIs and post-implementation monitoring
4. Implementation plan (how change rolls out safely)
- Staged rollout or pilot framing
- Training support (pharmacists)
- Communications plan (patients and healthcare providers)
- Metrics and evaluation timepoints
The consultation reality: public submissions matter
Scheduling changes are not made behind closed doors. The TGA publishers calls for submissions and summaries of scheduling matters, and the consultation inputs are considered alongside committee advice and delegate deliberations. [8, 14]
This is why stakeholder alignment is not “nice to have”—it is essential:
- Pharmacists (practical supply controls)
- Clinicians (clinical need and patient outcomes)
- Regulators (scheduling fit, enforceability)
- Public-health stakeholders (diversion risk management)
A pragmatic pathway forward: what “responsible reintroduction” could look like?
A credible, regulator-friendly approach is not “more access.” It is better-controlled access with better clinical outcomes.
A strong option to explore in policy terms is a monitored model that:
- Reinforces pharmacist oversight
- Relies on real-time recording systems already used to manage diversion risk
- Sets clear pack/quantity limits and counselling expectations
- Establishes measurable KPIs for safety and diversion outcomes
The key message is: Australia already built the infrastructure to manage risk—the scheduling framework exists to decide how to use it. [1, 2, 3]
Closing thought
If we want evidence-based OTC care, we need evidence-based scheduling conversations.
\The SUSMP framework supported by the Scheduling Policy Framework and Scheduling Handbook exists specifically to resolve tensions like this: benefit vs risk, access vs misuse potential. [1, 2, 3]
Pseudoephedrine’s question is no longer “does it work?”
It’s whether Australia can apply modern controls to restore effective patient outcomes while maintaining public safety.
References
[1] The Poisons Standard (the SUSMP)
[2] Scheduling Policy Framework for Medicines and Chemicals
[3] Scheduling handbook: Guidance for amending the Poisons Standard
[4] Advisory Committee on Medicines Scheduling (ACMS)
[5] Drugs and poisons scheduling
[6] Regulation impact statement: Codeine re-scheduling
[7] Scheduling committees meeting dates and decisions timeframes
[8] Public consultation on proposed amendments to the Poisons Standard – ACMS & Joint ACMS-ACCS
[9] Requirements for recording pseudoephedrine sales
[10] Project STOP A Pharmacy Guild initiative
[13] Regulation and Scheduling of Medicines and Poisons
Authors Note: Wilson Prasad also known as user name muefatiaki1966 is trying to leverage his experience working on products containing PSE in Australia for over 20 years and would like to express his interest and concern of PSE based products in Australia.
Leave a comment