As part of The Periodic Table article series, this article serves as a revision of the previous article, and provides more extensive detail compared to the previous.
Overview
| Property | Value |
| Symbol | Li |
| Atomic Number | 3 |
| Atomic Mass | 6.94 u |
| Group | 1 |
| Period | 2 |
| Classification | Alkali Metal |
| Electron Configuration | 1s² 2s¹ |
| State at 25°C | Solid |
Lithium is the lightest solid element and the first metal encountered in the periodic table.
Though relatively rare in the universe compared to hydrogen and helium, lithium occupies a unique scientific position: it bridges cosmology, geology, biology, and energy technology.
Lithium was formed during Big Bang nucleosynthesis, but in much smaller quantities than hydrogen and helium, making it an important probe for early universe physics. [1]
Today, lithium has become indispensable to modern society due to its central role in rechargeable batteries, enabling portable electronics, electric vehicles, and grid-scale energy storage.
Discovery and Naming
Lithium was discovered in 1817 by Johan August Arfwedson, a Swedish chemist working in the laboratory of Jöns Jakob Berzelius.
Arfwedson identified lithium while analysing the mineral petalite, noting a new alkali metal distinct from sodium and potassium. [2]
The name lithium is derived from the Greek word lithos, meaning stone, reflecting its mineral origin rather than discovery in plant or animal matter unlike other alkali metals known at the time.
Atomic Structure and Isotopes
Lithium atoms consist of:
- 3 protons
- 3 electrons
- Typically 3 or 4 neutrons
Lithium has two stable isotopes:
| Isotope | Symbol | Neutrons | Notes |
|---|---|---|---|
| Lithium-6 | ⁶Li | 3 | ~7.5% |
| Lithium-7 | ⁷Li | 4 | ~92.5% |
Lithium-6 is particularly important in nuclear fusion research, where it can produce tritium when bombarded with neutrons. [3]

Natural Occurrence and Extraction
Occurrence
Lithium is never found as a free element due to its high reactivity. It occurs in:
- Hard-rock minerals (E.g. Spodumene, petalite)
- Brine deposits (E.g. Salt lakes and underground reservoirs)
Major lithium resources are located in Australia, Chile, Argentina, and China. [4]
Extraction Methods
- Hard-rock mining followed by roasting and chemical processing
- Brine extraction using solar evaporation to concentrate lithium salts

Brine extraction currently dominates global supply due to lower energy requirements, though it raises environmental concerns regarding water use. [5]
Application and Uses of Lithium in Modern Technology
🔋 Energy Storage and Battery Technologies (Dominant Use)
Lithium’s low atomic mass and high electrochemical potential make it ideal for lithium-ion batteries.
Over 70% of global lithium production is now directed toward rechargeable battery technologies, particularly lithium-ion (Li-ion) batteries, which underpin modern electrification.
Used extensively in:
- Electric vehicles (EVs)
- Smartphones and laptops
- Grid-scale renewable energy storage
- Power tools and industrial robotics
Lithium-ion batteries exhibit high energy density, long cycle life, and relatively low self-discharge. [6]
Key lithium chemistries include:

- LFP (Lithium Iron Phosphate): safer, longer cycle life (e.g. Tesla standard-range EVs)
- NMC (Nickel-Manganese-Cobalt): high energy density (used in long-range EVs)
- NCA (Nickel-Cobalt-Aluminium): aerospace and premium EVs
Lithium’s role is critical in enabling fast charging, high energy density, and long operational life. [6, 7]
Solid-State Lithium Batteries (Next-Gen):
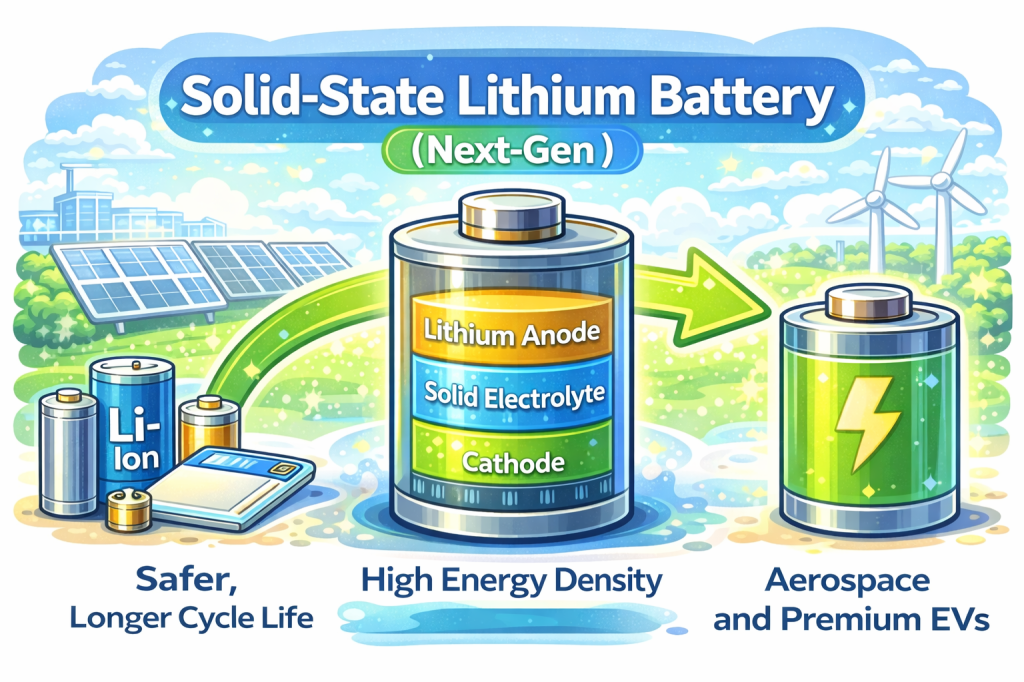
Solid-state batteries replace liquid electrolytes with solid lithium conductors, offering:
- Higher energy density
- Improved thermal safety
- Reduced fire risk
- Faster charging
Companies and research institutions actively developing this technology include:
Solid-state lithium batteries are expected to reach commercial deployment between 2026–2030. [9]
🧠 Medicine and Biology
Lithium salts, particularly lithium carbonate and lithium citrate, are widely prescribed for:
- Bipolar disorder [8]
- Mood stabilization
- Suicide risk reduction
Lithium is one of the few psychiatric medications with neuroprotective effects, influencing:
- Neurotransmitter signaling
- Neurogenesis
- Inflammation regulation
Emerging research is investigating lithium’s role in:
- Alzheimer’s disease
- Parkinson’s disease
- Traumatic brain injury recovery
Lithium is not considered an essential biological element, but trace amounts are present in natural waters and food sources. [8]
🧪 Industry and Materials Science
Glass and Ceramics:
Lithium compounds:
- Lower melting points
- Improve thermal shock resistance
- Enhance durability
Applications include:
- Laboratory glassware
- Smartphone screens
- Heat-resistant cookware
- Industrial ceramics
Aluminium-Lithium Alloys (Aerospace):
Lithium additions to aluminium:
- Reduce weight by up to 10%
- Increase stiffness and fatigue resistance

Used in:
- Aircraft fuselages
- Spacecraft structures
- Satellite frames
Major aerospace programs (Airbus, Boeing, SpaceX) utilize Al–Li alloys to improve fuel efficiency and payload capacity. [10]
🚀 Aerospace, Spaceflight & Defense
Lithium plays a strategic role in:
- Rocket propellants
- Thermal batteries
- High-energy density power systems
Lithium hydride and lithium aluminium hydride are used as:
- Hydrogen storage materials
- Reducing agents
- Lightweight shielding in nuclear systems
Lithium-based batteries are standard in:
- Satellites
- Space probes
- Military electronics
⚛️ Nuclear Energy & Fusion Research
Lithium-6 reacts with neutrons to produce tritium, a key fuel for fusion reactions:
⁶Li + n → ⁴He + ³H
Lithium is therefore essential in:
- Fusion blanket designs
- Experimental fusion reactors (ITER, DEMO)
Without lithium, sustained fusion power would be impossible. [3]
🌍 Grid-Scale Energy Storage & Renewable Integration
Lithium battery systems are widely deployed for:
- Grid frequency regulation
- Renewable energy smoothing (solar & wind)
- Peak demand management
Large-scale lithium battery installations now support:
- National grids
- Industrial microgrids
- Remote and island communities
Lithium-based storage is a cornerstone of net-zero energy strategies globally. [11]
♻️ Recycling, Circular Economy & Sustainability
As lithium demand accelerates, recycling has become critical:
- Hydrometallurgical recovery
- Direct cathode recycling
- Closed-loop battery supply chains
Modern recycling processes can recover:
- Lithium
- Cobalt
- Nickel
- Copper
Recycling is expected to supply 20–30% of lithium demand by 2035, reducing environmental and geopolitical risk. [12]

🔮 Emerging & Future Applications
Research frontiers include:
- Lithium–sulfur batteries (Li-S)
- Lithium–air batteries (Li–O₂)
- Lithium-based quantum materials
- Advanced hydrogen storage systems
Lithium remains one of the most research-intensive elements due to its central role in energy transformation.
Environmental and Sustainability Considerations
The rapid growth of lithium demand has raised concerns related to:
- Water consumption in arid regions
- Land disturbance and ecosystem impact
- Supply chain concentration
Recycling lithium from spent batteries and developing alternative chemistries are critical to long-term sustainability. [12]
Safety and Hazards
Metallic lithium:
- Reacts violently with water
- Can ignite in air
- Must be stored under oil or inert atmosphere
Lithium-ion batteries pose risks of thermal runaway if damaged or improperly managed, highlighting the importance of battery safety engineering. [7]
Scientific and Cultural Significance
- Lithium is one of only three elements formed in the Big Bang
- It plays a key role in validating cosmological models
- It underpins the global transition to electrification
Lithium has transformed from a geological curiosity into a strategic resource shaping the 21st-century energy landscape.
Related Elements
- Hydrogen (H): Lithium formation constrains Big Bang models
- Helium (He): Lithium is produced after helium in stellar evolution
- Sodium (Na): Next alkali metal in the periodic table
References
[1] The Primordial Lithium Problem
[2] The History, Relevance, and Applications of the Periodic System in Geochemistry
[3] Plasma physics and controlled nuclear fusion research 1980
[4] Mineral Commodity Summaries 2023 – LITHIUM Data Release
[6] Issues and challenges facing rechargeable lithium batteries
[7] The Li-Ion Rechargeable Battery: A Perspective
[8] Treatment of bipolar disorder
[9] A solid future for battery development
[10] Light Alloys
[11] Trends in batteries
[12] Recycling lithium-ion batteries from electric vehicles
Authors Note: Eddy Xu is trying to express his interest in STEM and provide educated articles to allow readers from all ages to learn science and tech. He has a strong passion to develop new learning methods to bridge the gap between education and industry practice in his journey.

Leave a comment