Continuing on from the previous article.
Medicines regulation exists to protect public health, but protection is incomplete when ineffective medicines dominate routine care.
Safety without efficacy is not neutral; it creates hidden costs for patients, pharmacists, clinicians, and health systems.
In Australia, the shift from Pseudoephedrine (PSE) to Phenylephrine (PE) as the default oral nasal decongestant was driven by diversion risk, not by clinical equivalence.
Nearly two decades later, regulatory frameworks, pharmacy controls, and monitoring systems have evolved but therapeutic standards have not been revisited.
The evidence now demands that they be.
Nasal congestion: a vascular condition with regulatory implications
Nasal congestion is primarily caused by vasodilation and venous engorgement of the nasal mucosa, leading to increased nasal airway resistance (NAR) and impaired airflow. [1]
This matters from a regulatory perspective because:
- Congestion relief is a pharmacodynamic endpoint, not a subjective marketing claim
- Effective treatment requires systemic α-adrenergic vasoconstriction
- Medicines that cannot achieve this mechanism cannot deliver meaningful benefit
Australia’s Scheduling Policy Framework requires regulators to assess “the purpose for which the substance is to be used and the extent of the benefit” alongside risk. [2]
If a medicine cannot deliver the intended physiological effect, that requirement is not met.
Pharmacokinetics: why oral exposure is decisive?
Pseudoephedrine (PSE)
Pseudoephedrine has:
- High oral bioavailability
- Predictable systemic exposure
- A consistent dose–response relationship
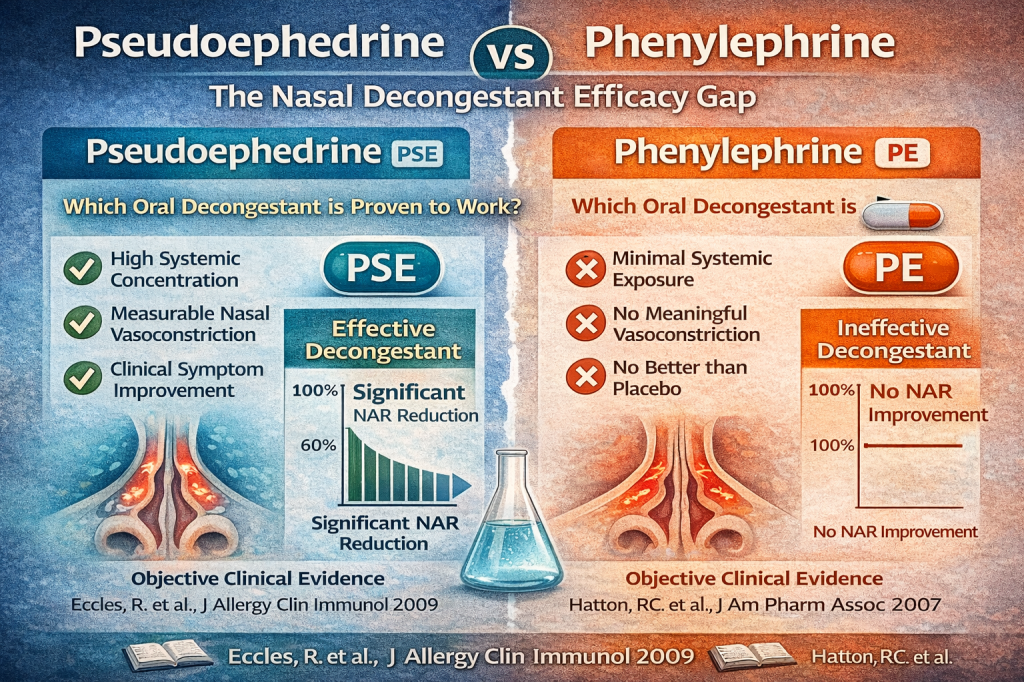
These properties allow PSE to reliably reach nasal vasculature and reduce NAR, which is why it has demonstrated consistent efficacy across controlled trials. [3, 4, 5]
Phenylephrine (PE)
Oral phenylephrine undergoes extensive first-pass metabolism, resulting in minimal systemic exposure.
Pharmacokinetic studies show that <1 % of an oral dose reaches circulation unchanged. [6]
From a regulatory science standpoint, this is critical:
A medicine with negligible systemic exposure cannot reasonably be expected to exert systemic nasal vasoconstriction.
This pharmacokinetic limitation directly undermines PE’s claimed therapeutic purpose.
Objective efficacy data: what controlled trials demonstrate
Subjective symptom scores are vulnerable to placebo effects. Regulatory science therefore prioritises objective measures such as NAR.
Pseudoephedrine (PSE)
Randomised, placebo-controlled trials consistently show that pseudoephedrine:
- Significantly reduces NAR
- Improves nasal airflow
- Produces clinically meaningful congestion relief
These findings are well documented in Journal of Allergy and Clinical Immunology and related challenge-chamber studies. [4, 5, 7]
Phenylephrine (PE)
In contrast, multiple controlled studies demonstrate that oral phenylephrine at standard OTC doses:
- Does not significantly reduce NAR
- Performs no better than placebo
- Shows no consistent dose–response effect
These findings are replicated across crossover trials, chamber studies, and comparative evaluations. [5, 8, 9, 10]
Systematic reviews: regulatory-grade evidence synthesis
Systematic and narrative reviews conclude that:
- Evidence supporting oral phenylephrine efficacy is weak or absent
- Positive studies are small, inconsistent, or methodologically limited
- Pseudoephedrine remains the only oral decongestant with robust, reproducible evidence
This distinction matters because regulators do not weigh isolated studies — they weigh bodies of evidence.
At this point, the evidence base for oral phenylephrine does not meet the threshold normally expected for therapeutic claims under modern regulatory scrutiny. [9, 10, 11]
International regulatory recognition
In 2023, the U.S. FDA Nonprescription Drugs Advisory Committee unanimously concluded that oral phenylephrine is ineffective at currently approved doses. [12]
This decision followed a comprehensive review of:
- Pharmacokinetics
- Pharmacodynamics
- Clinical trial data
- Real-world effectiveness
While FDA decisions are not binding in Australia, they are highly relevant comparator signals, particularly where:
- The pharmacology is universal
- The evidence base overlaps
- The clinical endpoint is identical
Australia’s Scheduling Policy Framework explicitly allows consideration of international regulatory experience where relevant to public health. [2]
Efficacy as a regulatory obligation (not a preference)
Under Australia’s scheduling framework, decision-makers must consider:
- Risks and benefits
- Extent of therapeutic use
- Toxicity and misuse potential
- Public-health impact
Efficacy is embedded in this framework — it is not optional.
An ineffective medicine can still cause harm by:
- Delaying effective treatment
- Increasing GP and emergency presentations
- Driving inappropriate antibiotic use
- Encouraging polypharmacy
- Undermining confidence in pharmacist-led care
These downstream effects are well documented in primary-care and respiratory medicine literature examining OTC treatment failure. [13]
The regulatory imbalance created by phenylephrine dominance
A credible, regulator-friendly approach is not “more access.” It is better-controlled Australia did not replace pseudoephedrine with a clinically equivalent alternative. It replaced it with a regulatorily convenient substitute.
That substitution:
- Reduced diversion risk under legacy systems
- But downgraded therapeutic outcomes
- And shifted the burden to pharmacists and patients
Today, Australia has:
- Real-time pseudoephedrine recording
- Pharmacist-only supply models
- Jurisdictional diversion controls
- Mature compliance frameworks
Yet efficacy has not been re-weighted accordingly.
The uncomfortable regulatory truth
Pseudoephedrine was restricted because systems were not ready to manage diversion risk — not because it lacked benefit.
Those systems now exist.
What has not yet occurred is a formal reassessment of efficacy within the scheduling framework, despite:
- Strong clinical evidence
- International regulatory signals
- Pharmacist experience
- Public health consequences of ineffective care
This creates a misalignment between regulatory intent and clinical reality.
Closing thought
Pseudoephedrine’s efficacy is not historical — it is current, reproducible, and well established.
What has expired is the assumption that phenylephrine is an adequate substitute.
Evidence-based regulation requires:
- Periodic reassessment
- Responsiveness to new data
- Alignment between safety, efficacy, and real-world outcomes
References
[1] Pathophysiology of Nasal Symptoms
[2] Scheduling Policy Framework for Medicines and Chemicals
[3] Martindale: The Complete Drug Reference
[6] Advocating for Patients Through Clinical Research
[8] Ambulatory Care: Efficacy and Safety of Oral Phenylephrine: Systematic Review and Meta-Analysis
[10] FDA clarifies results of recent advisory committee meeting on oral phenylephrine
[12] Final Summary Minutes of the Nonprescription Drugs Advisory Committee Meeting
Authors Note: Wilson Prasad also known as user name muefatiaki1966 is trying to leverage his experience working on products containing PSE in Australia for over 20 years and would like to express his interest and concern of PSE based products in Australia.
Leave a reply to The Pharmacist-Led Model: Restoring Access While Protecting Public Health – DeepDivers Cancel reply